COVID19
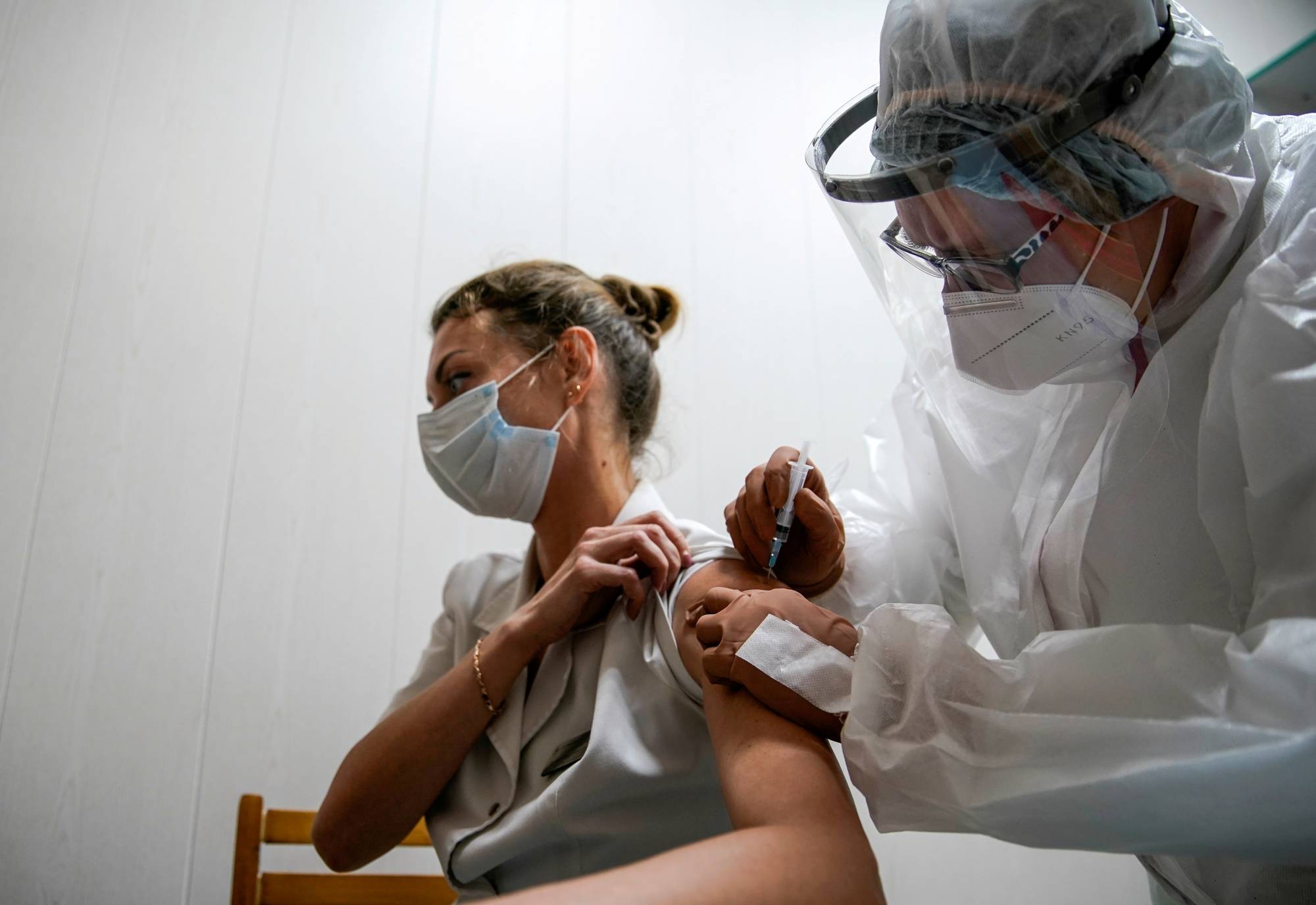
Dr Reddy’s records 5% surge in shares with roll out of Sputnik V COVID-19 vaccine
After various countries allowed the roll out of Sputnik V COVID-19 vaccine, Dr Reddy’s shares surged five per cent. Last month, Dr Reddy’s Laboratories received approval from the Drugs Control General of India (DCGI) to conduct phase 3 clinical trial for the Russian vaccine.
Its pharma stock, as per various reports, touched an intraday high of Rs 4,759.1 rising 6.13 per cent against previous close of Rs 4,484.25 on BSE. The share trades higher than 200 day, moving averages but lower than five day, 20 day, 50 day and 100 day moving averages.
As such, the stock has gained 48.49 per cent in one year and fallen 10.33 per cent since the beginning of this year. Market cap of the firm rose to Rs 77,601 crore. The stock has risen 5.01 per cent in the last two days.
Kirill Dmitriev, head of the Russian Direct Investment Fund (RDIF), which is responsible for marketing the vaccine abroad, said production has started in India and South Korea. He said Gamaleya Institute, which developed the vaccine, was testing the vaccine against new variants of COVID-19 and the early signs were positive. “India is a key partner in terms of production of the vaccine and we expect that there will be a restricted emergency use order for the Sputnik V vaccine this month or next. After that we will be able to start our vaccine delivery to India.”
Reports highlight Sputnik V vaccine as the fourth worldwide to have Phase III results published in leading peer-reviewed medical journals, following the shots developed by Pfizer and BioNTech, Moderna and AstraZeneca. Sputnik V was found to be 91.6 per cent effective against symptomatic COVID-19 cases in trial results published by The Lancet, a leading medical journal.
Also Read: Bharat Biotech hails Rs 35k cr budget allocation for COVID-19 vaccination
Dmitriev said the data published by The Lancet shows that Sputnik V is one of only three vaccines in the world with efficacy of over 90 per cent but outperforms them in terms of safety, ease of transportation due to storage requirements of two to eight degrees, and a more affordable price. He added that Sputnik V is a vaccine for all mankind.
Dr Reddy’s had partnered with the RDIF, in September 2020, to conduct the clinical trials of the Sputnik V vaccine and for its distribution rights in India.









































Pingback: Biden signs executive orders reversing Trump's hardline immigration policies | The Plunge Daily